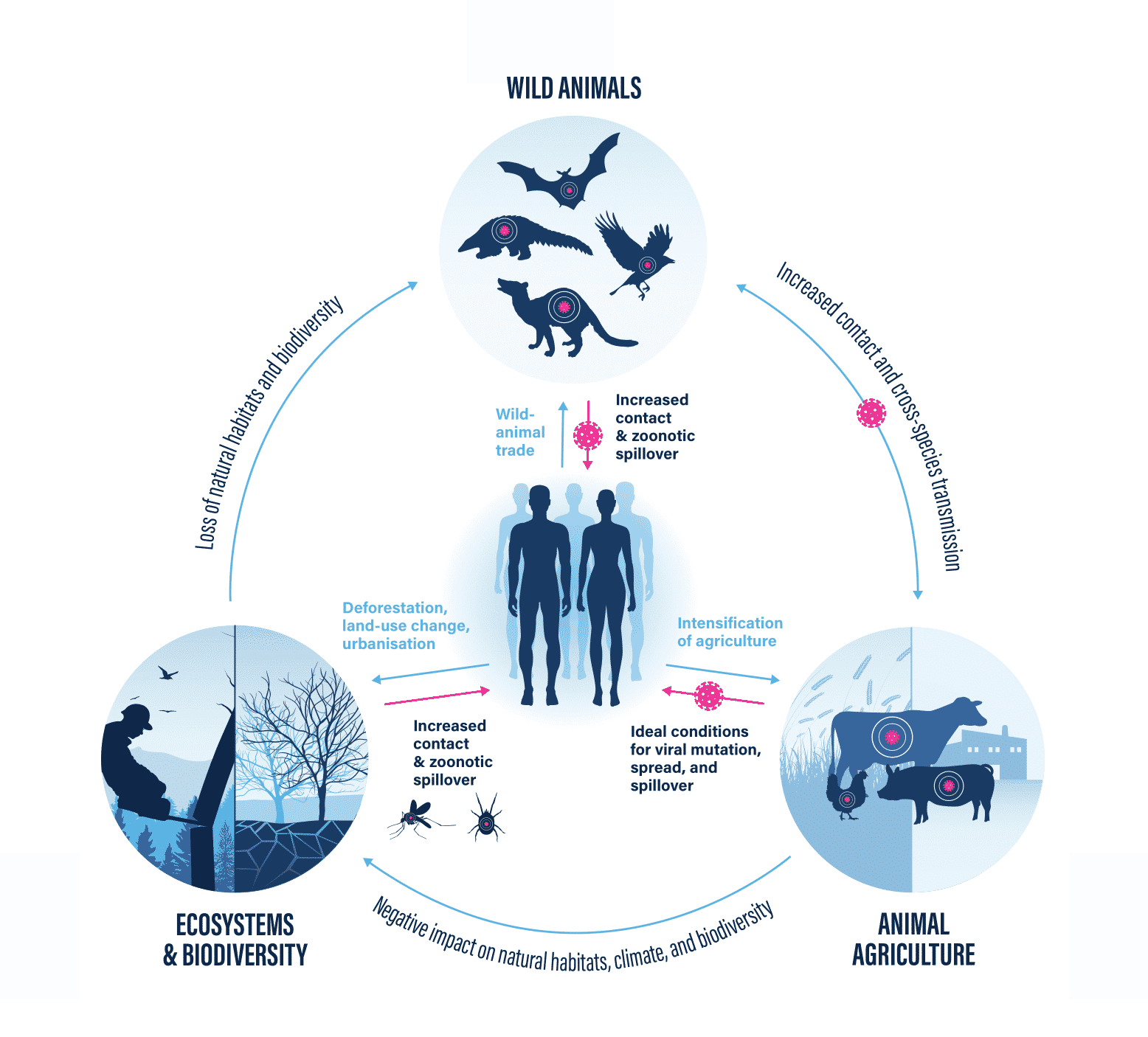
The emergence of a novel zoonotic disease is a highly complex process, involving many factors. Yet, there is compelling evidence that certain human activities strongly increase the likelihood of such developments. Three aspects of human behaviour are particularly devastating – all of which have to do with human-animal interactions in the context of food. In this blog, we’ll have a closer look at the first factor – The destruction of ecosystems and the loss of biodiversity (driven largely by animal agriculture) – resulting in increased contact and virus spillover to humans and farmed animals.
The trajectory of this interplay is alarming, given the world’s fast-growing appetite for animal protein, and intensified animal agriculture’s key role in satisfying it. With meat and milk production expected to grow by approximately 15% by 20281 due to rising global human population and prosperity levels, future pandemic risk is also expected to further increase.
Moving away from animal agriculture and animal-based products can help preserve ecosystems and biodiversity, reduce interference with wild animal species, and remove the need for factory farms that provide hotbeds for zoonotic pandemic emergence and spread. Shifting to a better, more resilient, and sustainable global food system that replaces animal products with plant-based and cultured alternatives ranks among the best options. It provides a multi problem solution that not only mitigates future pandemic risks, but also helps to minimise major parallel crises such as climate change, world hunger, and antibiotic resistance.
THE DESTRUCTION OF ECOSYSTEMS AND LOSS OF BIODIVERSITY
The health of humans is directly linked to the health of the planet and all its inhabitants. While most research is clear about the importance of intact ecosystems, human activities don’t seem to reflect this insight. Instead, there is an ever-increasing exploitation and disruption of ecosystems, causing species diversity and animals’ natural habitats to continuously decline.
The sixth mass extinction – human-made biodiversity loss
We are living in the midst of the sixth mass extinction on the planet and are facing a rapid global loss of biodiversity. For the first time in the history of the planet, such an event is human-made.2 Our actions have heavily impacted more than 75% of the Earth’s land surface,3 significantly altering the prevalence and composition of its flora and fauna. Expanding urbanisation, as well as increasing commercial and infrastructural activities, driven by growing populations and prosperity levels, are significant contributors to these developments4 – with animal agriculture playing a central role. Tropical regions with high biodiversity are particularly vulnerable to, and affected by, all of these developments. The consequences are dramatic: more encroachment into, and destruction of, natural ecosystems means greater exposure to hitherto unknown viruses – and an increased risk of zoonotic spillover.5 6 7
Animal agriculture – a key driver of the destruction of ecosystems
Animal agriculture is one of the key drivers of land-use change worldwide, as forests are cleared to provide space for feed crops and pastures in order to supply the increasing demand for meat.8 9 10 11 12 13 Currently, 70% of fresh water and 50% of habitable land is used for crops and livestock production,14 15 while more than 80% of the world’s farmland is used for the production of meat, eggs, and dairy. However, animal-based products provide a mere 18% of global calories.16
Compared to other forms of agriculture, livestock farming is particularly inefficient owing to its poor protein-efficiency ratio (that is, the quantity of plant protein required to produce one kilogramme of edible animal protein). Farmed animals need to consume between 6 and 12.5 kg of plant protein in order to produce a single kilogram of animal protein.17 18 19 The consumption of animal-based products leads to massive encroachments into natural habitats – with very low yields.20 21
Decreased biodiversity and increased zoonotic emergence
There is now substantial evidence that shows a clear correlation between human intrusion into ecosystems and the resultant habitat destruction, and an increased risk of pathogenic spillover. Ecosystems consist of communities of plants, animals, and microorganisms, as well as all the physical and chemical components of a specific environment or habitat. The interactions between all the components of an ecosystem are highly complex. As are the effects of biodiversity loss on pathogens, since their biological life cycles, as well as climate and host requirements, may vary greatly.22 While some pathogens might be very specialised in relation to a specific host, there are others that have a larger host range and might be able to cross species barriers more easily.
Undisturbed habitats allow for a natural composition and balanced spatial distribution of species, which can result in a high diversity of both animals and pathogens.23 While a high diversity of pathogens sounds like a problem, this seems not to be the case: the dilution-effect hypothesis proposes that undisturbed habitats with high biodiversity make it much more difficult for pathogens such as viruses to spread and find suitable hosts.24 25 26 27 28
The dilution effect – nature’s version of social distancing
One of the reasons for this is that the more diverse a biological community, the higher the probability is that there are species and individuals that are immune to a virus or unsuitable as a host. In habitats with high biodiversity, the number of individuals of the same species within a population may be lower (owing to natural regulating mechanisms such as predation or competition between species). As a consequence, viruses spread more slowly or are stopped by natural barriers. In other words, the risk of pathogens spreading is ‘diluted’ – a little like nature’s version of social distancing. However, if a habitat contains only a few animal species that are potential hosts for viruses and those animal species are genetically very similar (for example as a consequence of human environmental interference), then the virus can spread easily. And it might become so abundant that there is an increased risk that it will evolve the ability to jump the species barrier and infect other species – including humans.29 30
Loss of diversity + increase in numbers = higher spillover risk
This is supported by evidence that mammal species (such as rodents, bats, primates, and domesticated animals) that have increased in population size due to human environmental interference share more viruses with humans than less abundant species. Human interference with the composition and numbers of wild animals thus increases the chances of interaction between wild animal species – and the viruses they carry – who would never meet under natural conditions in intact ecosystems. Adding domesticated animals such as farmed animals to this equation further increases the risk of pathogen transmission – making a zoonotic spillover event to humans significantly more likely.31
Climate change – a risk multiplier
Along with human population growth and the anthropogenic destruction of ecosystems, climate change also contributes to the increased transmission of pathogens from host animals to humans.32 Changing climatic conditions such as higher average temperatures can shift the habitats of both animals and pathogens.33 34 35 This further spurs imbalances in ecosystems and loss of biodiversity, resulting in higher risks of new infectious diseases emerging.36 37 In addition, changes to the climate may also favour the spread of vector-borne diseases (diseases that are transmitted by carriers such as ticks and fleas). Alarming examples include malaria or dengue fever, which are now expanding to new geographical areas, since the mosquitoes which transmit them are finding favourable conditions in these areas.38 39 40 41 There is no doubt that climate change is the result of human activities. One of the main drivers of climate change is animal agriculture, accounting for about 16% of global greenhouse gas emissions,42 while also being a major contributor to environmental degradation.
A destructive interplay – fuelled by our food choices
Our environmentally destructive activities bring us closer to wild animals and their often unknown pathogens. This creates favourable conditions for viruses to spread, and ultimately enables the development of dangerous zoonoses that have the potential to become global pandemics. Add increasing temperatures and changing climate conditions to the equation, and the threat multiplies substantially.
Profound impacts – and little awareness
Although there is widespread agreement about the destructive effects of this interplay, there is alarmingly little awareness of the fact that animal agriculture is one of its key drivers. While stressing the need to preserve ecosystems, natural habitats, and biodiversity, even experts often fail to make the connection. This is particularly unfortunate in light of the fact that animal agriculture contributes substantially to both environmental destruction and climate change. Globally, we raise and kill more than 75 billion land animals every year,43 with that number continually increasing. Breeding, feeding, processing, and shipping these staggering numbers of animals takes up vast resources, including land and water, and consequently leads to massive impacts on global ecosystems.
In order to address the occurrence and transmission of pathogens and the emergence of future zoonotic pandemics, we need to address our preference for animal-based foods – including both the products of animal agriculture and using wild animals as food.
Wild animals as food – increasing the risk of zoonotic pandemics
By exploring the crucial connection between the current crisis and our animal-based food system, the ProVeg Food & Pandemics Report highlights how our food choices help to create a recipe for zoonotic pandemics.
References
- OECD & FAO (2019): OECD-FAO Agricultural Outlook 2019-2028. OECD Publishing, Paris/ Food and Agriculture Organization of the United Nations, Rome. doi:10.1787/agr_outlook-2019-en doi:10.1787/agr_outlook-2019-en
- Ceballos, G., P. R. Ehrlich & R. Dirzo (2017): Biological annihilation via the ongoing sixth mass extinction signaled by vertebrate population losses and declines. Proceedings of the National Academy of Sciences 114(30), E6089–E6096. doi:10.1073/pnas.1704949114
- IPBES (2018): The IPBES assessment report on land degradation and restoration. Montanarella, L., Scholes, R., and Brainich, A. (eds.). Secretariat of the Intergovernmental Science-Policy Platform on Biodiversity and Ecosystem Services. Bonn, Germany. p.69
- Machovina, B., K. J. Feeley & W. J. Ripple (2015): Biodiversity conservation: The key is reducing meat consumption. Science of The Total Environment 536 419–431. doi:10.1016/j.scitotenv.2015.07.022
- Olival, K. J., P. R. Hosseini, C. Zambrana-Torrelio, et al. (2017): Host and viral traits predict zoonotic spillover from mammals. Nature 546(7660), 646–650. doi:10.1038/nature22975
- Jones, K.E., N.G. Patel, M.A. Levy et al. (2008): Global trends in emerging infectious diseases. Nature, 451, 990-993. https://doi.org/10.1038/nature06536
- Hassell, J. M., M. Begon, M. J. Ward, et al. (2017): Urbanization and Disease Emergence: Dynamics at the Wildlife–Livestock–Human Interface. Trends in Ecology & Evolution32(1), 55–67. doi:10.1016/j.tree.2016.09.012
- Kissinger, G., M. Herold & De Sy, Veronique (2012): Drivers of Deforestation and Forest Degradation: A Synthesis Report for REDD+ Policymakers. Lexeme Consulting, Vancouver Canada
- FAO (2018): The State of the World’s Forests 2018 – Forest pathways to sustainable development. FAO, Rome
- Curtis, P. G., C. M. Slay, N. L. Harris, et al. (2018): Classifying drivers of global forest loss. Science 361(6407), 1108–1111. doi:10.1126/science.aau3445
- Poore, J. & T. Nemecek (2018): Reducing food’s environmental impacts through producers and consumers. Science 360(6392), 987–992. doi:10.1126/science.aaq0216
- Maxwell, S. L., R. A. Fuller, T. M. Brooks, et al. (2016): Biodiversity: The ravages of guns, nets and bulldozers. Nature 536(7615), 143–145. doi:10.1038/536143a
- Machovina, B., K. J. Feeley & W. J. Ripple (2015): Biodiversity conservation: The key is reducing meat consumption. Science of The Total Environment 536 419–431. doi:10.1016/j.scitotenv.2015.07.022
- UNESCO & UN-Water (2020): United Nations World Water Development Report 2020: Water and Climate Change. UNESCO, Paris. p. 5
- Ritchie, H. & M. Roser (2019): Land Use. Our World in Data. Available at https://ourworldindata.org/land-use. [Accessed: 22.5.2020]
- Poore, J. & T. Nemecek (2018): Reducing food’s environmental impacts through producers and consumers. Science 360(6392), 987–992. doi:10.1126/science.aaq0216
- Pimentel, D. & M. Pimentel (2003): Sustainability of meat-based and plant-based diets and the environment. The American Journal of Clinical Nutrition 78(3), Oxford Academic, 660S-663S. doi:10.1093/ajcn/78.3.660S
- Boyd, C. E. Protein conversion efficiency in aquaculture « Global Aquaculture Advocate. Global Aquaculture Alliance. Available at https://www.aquaculturealliance.org/advocate/protein-conversion-efficiency-in-aquaculture/. [Accessed: 20.5.2020]
- Shepon, A., G. Eshel, E. Noor, et al. (2016): Energy and protein feed-to-food conversion efficiencies in the US and potential food security gains from dietary changes. Environmental Research Letters 11(10), IOP Publishing, 105002. doi:10.1088/1748-9326/11/10/105002
- Alexander, P., C. Brown, A. Arneth, et al. (2016): Human appropriation of land for food: The role of diet. Global Environmental Change. 41, p.88–98
- Shepon, A., G. Eshel, E. Noor, et al. (2018): The opportunity cost of animal based diets exceeds all food losses. PNAS. 115, National Academy of Sciences p.3804–3809
- Rohr, J. R., D. J. Civitello, F. W. Halliday, et al. (2020): Towards common ground in the biodiversity–disease debate. Nature Ecology & Evolution 4(1), 24–33. doi:10.1038/s41559-019-1060-6
- Mollentze, N. & D. G. Streicker (2020): Viral zoonotic risk is homogenous among taxonomic orders of mammalian and avian reservoir hosts. Proceedings of the National Academy of Sciences 117(17), 9423–9430. doi:10.1073/pnas.1919176117
- Keesing, F., L. K. Belden, P. Daszak, et al. (2010): Impacts of biodiversity on the emergence and transmission of infectious diseases. Nature 468(7324), 647–652. doi:10.1038/nature09575
- Khalil, H., F. Ecke, M. Evander, et al. (2016): Declining ecosystem health and the dilution effect. Scientific Reports 6(1), doi:10.1038/srep31314
- Rohr, J. R., D. J. Civitello, F. W. Halliday, et al. (2020): Towards common ground in the biodiversity–disease debate. Nature Ecology & Evolution 4(1), 24–33. doi:10.1038/s41559-019-1060-6
- Civitello, D. J., J. Cohen, H. Fatima, et al. (2015): Biodiversity inhibits parasites: Broad evidence for the dilution effect. Proceedings of the National Academy of Sciences 112(28), 8667–8671. doi:10.1073/pnas.1506279112
- Rohr, J. R., D. J. Civitello, F. W. Halliday, et al. (2020): Towards common ground in the biodiversity–disease debate. Nature Ecology & Evolution 4(1), 24–33. doi:10.1038/s41559-019-1060-6
- King, K. C. & C. M. Lively (2012): Does genetic diversity limit disease spread in natural host populations? Heredity 109(4), 199–203. doi:10.1038/hdy.2012.33
- Parrish, C. R., E. C. Holmes, D. M. Morens, et al. (2008): Cross-Species Virus Transmission and the Emergence of New Epidemic Diseases. Microbiology and Molecular Biology Reviews 72(3), 457–470. doi:10.1128/MMBR.00004-08
- Johnson, C. K., P. L. Hitchens, P. S. Pandit, et al. (2020): Global shifts in mammalian population trends reveal key predictors of virus spillover risk. Proceedings of the Royal Society B: Biological Sciences 287(1924), 20192736. doi:10.1098/rspb.2019.2736
- Redding, D. W., L. M. Moses, A. A. Cunningham, et al. (2016): Environmental-mechanistic modelling of the impact of global change on human zoonotic disease emergence: a case study of Lassa fever. Methods in Ecology and Evolution 7(6), 646–655. doi:10.1111/2041-210X.12549
- Mills, J. N., K. L. Gage & A. S. Khan (2010): Potential Influence of Climate Change on Vector-Borne and Zoonotic Diseases: A Review and Proposed Research Plan. Environmental Health Perspectives 118(11), 1507–1514. doi:10.1289/ehp.090138
- Slenning, B. D. (2010): Global Climate Change and Implications for Disease Emergence. Veterinary Pathology 47(1), 28–33. doi:10.1177/0300985809354465
- Ogden, L. E. (2018): Climate change, pathogens, and people. BioScience 68(10), 733–739. doi:10.1093/biosci/biy101
- WHO (2018): Climate change and health. World Health Organization. Available at https://www.who.int/news-room/fact-sheets/detail/climate-change-and-health. [Accessed: 20.5.2020]
- Jordan, R. (2019): How does climate change affect disease? Stanford University – Stanford Earth School of Earth, Energy & Environmental Sciences. Available at https://earth.stanford.edu/news/how-does-climate-change-affect-disease. [Accessed: 20.4.2020]
- Wu, X., Y. Lu, S. Zhou, et al. (2016): Impact of climate change on human infectious diseases: Empirical evidence and human adaptation. Environment International 86 14–23. doi:10.1016/j.envint.2015.09.007
- Peters, A., P. Vetter, C. Guitart, et al. (2020): Understanding the emerging coronavirus: what it means for health security and infection prevention. Journal of Hospital Infection 104(4), 440–448. doi:10.1016/j.jhin.2020.02.023
- Semenza, J. C. & J. E. Suk (2018): Vector-borne diseases and climate change: a European perspective. FEMS Microbiology Letters 365(2), doi:10.1093/femsle/fnx24
- Caminade, C., K. M. McIntyre & A. E. Jones (2019): Impact of recent and future climate change on vector‐borne diseases. Annals of the New York Academy of Sciences 1436(1), 157–173. doi:10.1111/nyas.13950
- Based on FAO (2017): Global Livestock Environmental Assessment Model (GLEAM). Rome. Available at: http://www.fao.org/gleam/results/en/ and IPCC (2014): Climate Change 2014: Synthesis Report. Contribution of Working Groups I, II and III to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change [Core Writing Team, R.K. Pachauri and L.A. Meyer (eds.)]. IPCC, Geneva, Switzerland
- Food and Agriculture Organization of the United Nations (2020): Livestock primary. FAOSTAT Database. Rome, Italy. Available at: http://www.fao.org/faostat/en/#data/QL [Accessed: 26.06.2020]










